–{•¶‚إ‹cک_‚³‚ê‚é–Fچپ‘°ƒJƒ‹ƒ{ƒ“ژ_‚جچ\‘¢
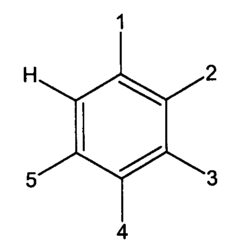
** = represents double bond.
*** R1 represents CH2C[OH][COOH]CH[OH]COOH where [ ] represents branched group.
پwAbstract
پ@Concentrations of organic acids in prebiotic soils were presumably
low, given limitations in abiotic synthesis and the limited lifetimes
of organic molecules before the ultraviolet shield developed on
early Earth. Prokaryotes, the first land-colonizing organisms,
commonly secrete aliphatic carboxylic acids, and, less extensively,
secrete aromatic compounds as siderophores and antibiotic. In
contrast, secretion of aromatic acids is considerable for fungi,
lichens, and vascular plants. Aromatic acids are also produced
by degradation oh high-molecular-weight compounds from lignin
and tannin, both abundant in vascular plants. The proportion of
aromatic carboxylic acids in soil solutions therefore probably
increased with the evolution of higher order organisms. As biomass
of organisms increased over geological time, concentrations of
organic acids in soil solutions and, in turn, the extent of ligand-promoted
dissolution of minerals probably increased.
پ@To elucidate the contribution of ligands during weathering on
early Earth, Columbia River basalt was dissolved under oxic and
anoxic conditions in the presence (0.001 or 0.01 M) and absence
of several organic ligands in batch experiments at pH 6. Release
of all elements including Si was enhanced considerably in the
presence of organic ligands. Citrate (tridentate) and gallate
(tetradentate) increased element release to the greatest extent
among the aliphatic and aromatic ligands, respectively. The extent
of element mobilization observed for the aliphatic ligands
decreased in the order: citrateپ„oxalateپàmalonate, and for
the aromatic ligands: gallateپ„salicylateپàphthalate.
The effects of the ligands generally followed trends in cation-ligand
stability constants, but aromatic ligands were less effective
in element mobilization than aliphatic ligands. One exception
was gallate, an aromatic ligand, which significantly enhanced
Cu release. Ligand-promoted mobilization of Cu may therefore have
increased over geologic time with the increase in the proportion
of aromatic ligands.
پ@In the presence of organic ligands, Fe was mobilized from basalt
considerably more than Al even under oxic conditions. Complexation
of Fe with organic ligands may have mobilized Fe in Precambrian
paleosols where little Al mobility is observed. Extent of P and
Y release was minor in ligand-free experiments and considerable
with ligands regardless of PO2.
Release of Cu was considerable under oxic conditions, especially
with ligands, and minor under anoxic conditions. Mobility patterns
of P and Y could thus possibly serve as پgorganomarkersپh (indicative
of prevalence of organic ligands in soil solutions) and mobility
patterns of Cu could possibly serve as پgoxymarkersپh (indicative
of the presence of molecular oxygen), respectively, in ancient
soils.پx
Introduction
Evolution of moieties of organic acids
پ@Abiotic synthesis
پ@General overview of biological evolution
پ@Secretion of LMWOA by Prokaryotes
پ@Secretion of LMWOA by fungi and lichens
پ@LMWOA in plant root exudates
پ@LMWOA as degradation products of HMW compounds
پ@Summary of the evolution of organic acid moieties
Statement of objectives
Materials and methods
پ@Organic ligands
پ@Starting materials
پ@Batch dissolution experiments
Results and discussion
پ@Solution chemistry
پ@Rate of dissolution
پ@Ligand trends
پ@Mobility of Si
پ@Order of element release
پ@Zr mobility
پ@Implications of Fe and Al mobility patterns
پ@Interpretations of P, Y and Cu mobility patterns
پ@Cu, P, and Y in paleosols
پ@Cu mobility over geologic time
Conclusions
Acknowledgments
Appendix 1
Appendix 2
References
|
|
|
|
|
| Acetic | گ|ژ_ | CH3COOH | 2 |
| Aconitic | ƒAƒRƒjƒbƒgژ_ | HOOCC=[CHCOOH]CH2COOH | 6 |
| Butyric | —ڈژ_ | CH3(CH2)2COOH | 4 |
| Citric | ƒNƒGƒ“ژ_ | HOC[CH2COOH]2COOH | 6 |
| Formic | ƒMژ_ | HCOOH | 1 |
| Fumaric | ƒtƒ}ƒ‹ژ_ | HOOCCH=CHCOOH | 4 |
| Glutaric | ƒOƒ‹ƒ^ƒ‹ژ_ | HOOC(CH2)3COOH | 5 |
| Glycolic | ƒOƒٹƒRپ[ƒ‹ژ_ | HOCH2COOH | 2 |
| Glyoxylic | ƒOƒٹƒIƒLƒVƒ‹ژ_ | OCHCOOH | 2 |
| Isocitric | ƒCƒ\ƒNƒGƒ“ژ_ | HOOCCH2CH[COOH]CH[COOH]OH | 6 |
| 2-ketogluconic | 2-ƒPƒgƒOƒ‹ƒRƒ“ژ_ | HOOCC[O](CH[OH])3CH2OH | 6 |
| ƒ؟-ketoglutaric | ƒ؟-ƒPƒgƒOƒ‹ƒ^ƒ‹ژ_ | HOOC(CH2)2C[O]COOH | 5 |
| Lactic | “ûژ_ | CH3CH[OH]COOH | 3 |
| Malic | ƒٹƒ“ƒSژ_ | HOOCCH2CH[OH]COOH | 4 |
| Malonic | ƒ}ƒچƒ“ژ_ | HOOCCH2COOH | 3 |
| Oxalic | ƒVƒ…ƒEژ_ | HOOCCOOH | 2 |
| Oxaloacetic | ƒIƒLƒTƒچگ|ژ_ | HOOCC[O]CH2COOH | 4 |
| Propionic | ƒvƒچƒsƒIƒ“ژ_ | CH3CH2COOH | 3 |
| Pyruvic | ƒsƒ‹ƒrƒ“ژ_ | CH3C[O]COOH | 3 |
| Succinic | ‚±‚ح‚ژ_ | HOOC(CH)2COOH | 4 |
| Tartaric | ژًگخژ_ | HOOC(CH[OH])2COOH | 4 |
| Valeric | ‹g‘گژ_ | CH3(CH2)3COOH | 5 |
| * [ ] represents branched group and = represents double bond. | |||
 |
||||||
|
|
|
|
||||
| 1 | 2 | 3 | 4 | 5 | ||
| Benzoic | ˆہ‘§چپژ_ | COOH | H | H | H | H |
| Caffeic | ƒRپ[ƒqژ_پiƒJƒtƒFپ[ژ_پj | CH=CHCOOH | H | OH | OH | H |
| Cinnamic | “÷Œjژ_پiŒj”çژ_پj | CH=CHCOOH | H | H | H | H |
| p-Coumaric | p-ƒNƒ}ƒ‹ژ_ | CH=CHCOOH | H | H | OH | H |
| Ferulic | ƒtƒFƒ‹ƒ‰ژ_ | CH=CHCOOH | H | OCH3 | OH | H |
| Gallic | –vگHژqژ_ | COOH | H | OH | OH | OH |
| p-Hydrobenzoic | p-ƒqƒhƒچˆہ‘§چپژ_ | COOH | H | H | OH | H |
| Phthalic | ƒtƒ^ƒ‹ژ_ | COOH | COOH | H | H | H |
| Piscidic | پ@ | R1*** | H | H | OH | H |
| Salicylic | ƒTƒٹƒ`ƒ‹ژ_ | COOH | OH | H | H | H |
| Syringic | پ@ | COOH | H | OCH3 | OH | OCH3 |
| Terephthalic | ƒeƒŒƒtƒ^ƒ‹ژ_ | COOH | H | H | COOH | H |
| Vanillic | ƒoƒjƒٹƒ“ژ_ | COOH | H | OCH3 | OH | H |
|
* The structures of lichen acids (polyphenolic compounds)
are complex and thus are not shown. They are discussed, for example,
in Huneck and Yoshimura (1996). ** = represents double bond. *** R1 represents CH2C[OH][COOH]CH[OH]COOH where [ ] represents branched group. |
||||||