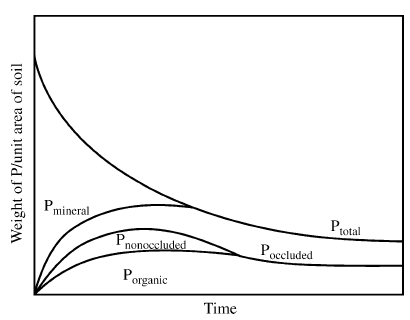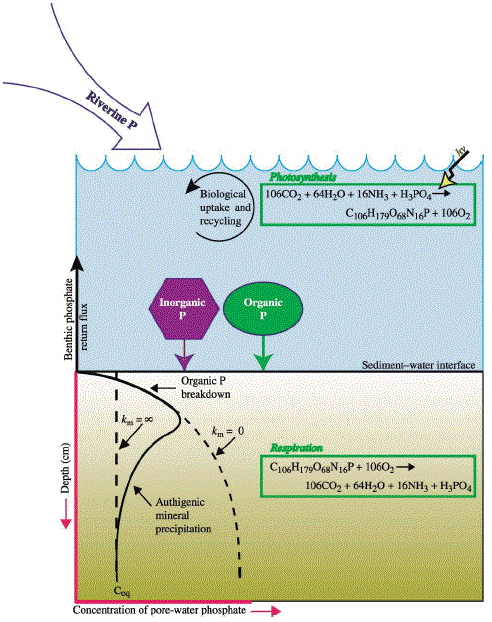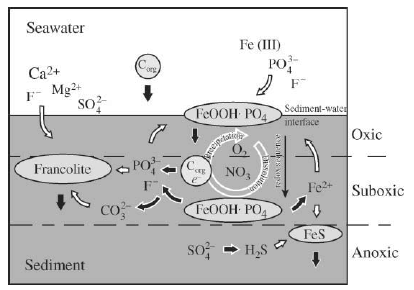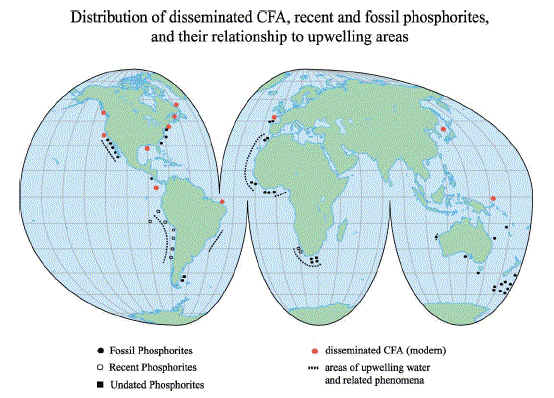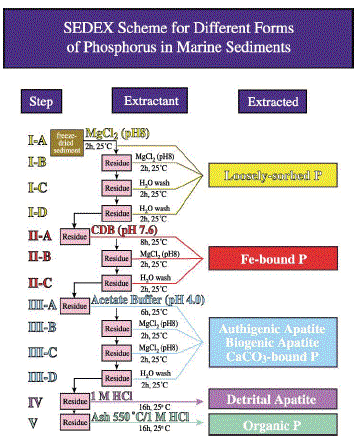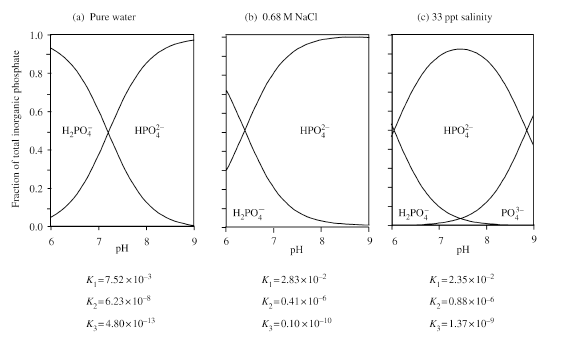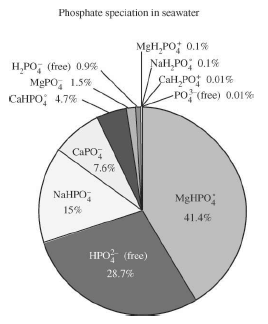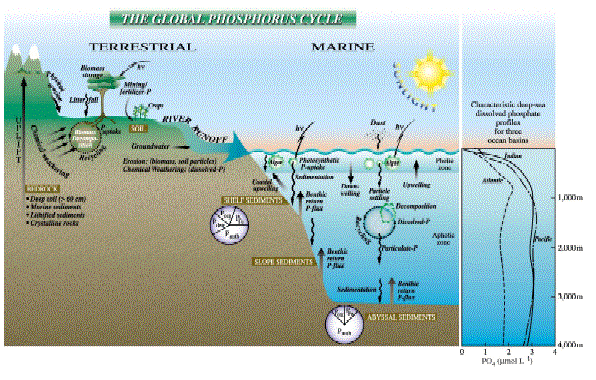
Figure 1 The major reservoirs and fluxes of the global phosphorus cycle are illustrated (see Tables 1 and 2, and text). The oceanic photic zone, idealized in the cartoon, is typically thinner in coastal environments due to turbidity from continental terrigenous input, and deepens as the water column clarifies with distance away from the continental margins. The distribution of phosphorus among different chemical/mineral forms in marine sediments is given in the pie diagrams, where the abbreviations used are: organic phosphorus (Porg), iron-bound phosphorus (PFe), detrital apatite (Pdetr), authigenic/biogenic apatite (Pauth). The Porg, PFe, and Pauth reservoirs represent potentially reactive-P pools (see text and Tables 2 and 3 for discussion), whereas the Pdetr pool reflects mainly detrital apatite weathered off the continents and passively deposited in marine sediments (note that Pdetr is not an important sedimentary phosphorus component in abyssal sediments, far from continents). Continental marginپ@P-speciation data were compiled from Louchouarn et al. (1997), and Ruttenberg and Berner (1993). Abyssal sediment P-speciation data were compiled from Filippelli and Delaney (1996), and Ruttenberg (1990). The پgglobal phosphorus cycleپh cartoon is from Ruttenberg (2002). The vertical water column phosphate distributions typically observed in the three ocean basins are shown in the panel to the right of the پgglobal phosphorus cycleپh cartoon, and are from Sverdrup et al. (1942).
Ruttenberg,K.C.(2003)‚ة‚و‚éپw8.13 The global phosphate cycleپx‚©‚ç
گ}‚Pپ@گ¢ٹE‚جƒٹƒ“ڈzٹآ‚جژه‚بƒٹƒUپ[ƒo‚ئƒtƒ‰ƒbƒNƒX‚ھگ}ژ¦‚³‚ê‚ؤ‚¢‚éپi•\‚P‚ئ•\‚Q‚ئ–{•¶‚ًژQڈئپjپBٹC—m—LŒُ‘w‘ر‚حپAگ}‚إ‚ح—‘z‰»‚³‚ê‚ؤ‚¢‚é‚ھپA‘ه—¤‚جچس‹û•¨‚ج—¬“ü‚ة‚و‚éچ¬‘÷‚ة‚و‚ء‚ؤ‰ˆٹفٹآ‹«‚إ‚ح“TŒ^“I‚ة”–‚‚ب‚ء‚ؤ‚¨‚èپA‘ه—¤‰ڈ‚©‚ç—£‚ê‚ؤگ…’Œ‚ھگں‚ق‚ة‚آ‚ê‚ؤگ[‚‚ب‚éپBٹC—m‘حگد•¨’†‚جˆظ‚ب‚鉻ٹw“Iپ^چz•¨ٹw“IŒ`‘شٹش‚إ‚جƒٹƒ“‚ج•ھ•z‚ح‰~ƒOƒ‰ƒt‚إ—^‚¦‚ؤ‚ ‚èپAژg—p‚³‚ê‚ؤ‚¢‚é—ھڈج‚حژں‚ج‚ئ‚¨‚èپF—L‹@ƒٹƒ“پiPorgپjپA“SŒ‹چ‡ƒٹƒ“پiPFeپjپAچس‹ûƒAƒpƒ^ƒCƒgپiPdetrپjپAژ©گ¶پ^گ¶•¨Œ¹ƒAƒpƒ^ƒCƒgپiPauthپjپBPorg‚ئPFe‚ئPauth‚جƒٹƒUپ[ƒo‚حگِچف“I‚ة”½‰گ«P‚جƒvپ[ƒ‹‚ًژ¦‚µپi‹cک_‚ح–{•¶‚¨‚و‚ر•\‚Q‚ئ•\‚R‚ًژQڈئپjپAPdetr‚جƒvپ[ƒ‹‚حژه‚ة‘ه—¤‚©‚ç•—‰»‚إ—¬ڈo‚µ‚½چس‹ûƒAƒpƒ^ƒCƒg‚إ‚ ‚èپAٹC—m‘حگد•¨’†‚ةژَ“®“I‚ة‘حگد‚µ‚½پiPdetr‚حپA‘ه—¤‚©‚ç—£‚ꂽ‰“—m‘حگد•¨’†‚جڈd—v‚ب‘حگدƒٹƒ“گ¬•ھ‚إ‚ح‚ب‚¢‚±‚ئ‚ة’چˆسپjپB‘ه—¤‰ڈ‚جP‰»ٹwژيƒfپ[ƒ^‚حLouchouarn et al.(1997)‚¨‚و‚رRuttenberg and Berner(1993)‚©‚ç‚ـ‚ئ‚ك‚½پB‰“—m‘حگدP‰»ٹwژيƒfپ[ƒ^‚حFilippelli and Delaney(1996)‚¨‚و‚رRuttenberg(1990)‚©‚ç‚ـ‚ئ‚ك‚½پBپwگ¢ٹE‚جƒٹƒ“ڈzٹآپx‚جگ}‚حRuttenberg(2002)‚©‚ç‚ج‚à‚ج‚إ‚ ‚éپB3‘هٹC–~‚إ“TŒ^“I‚ةٹد‘ھ‚³‚ꂽگ‚’¼گ…’Œƒٹƒ“•ھ•z‚حپwگ¢ٹE‚جƒٹƒ“ڈzٹآپx‚جگ}‚ج‰E‚ةƒpƒlƒ‹‰و‚ئ‚µ‚ؤژ¦‚³‚ê‚ؤ‚¢‚é‚ھپASverdrup et al.(1942)‚©‚ç‚ج‚à‚ج‚إ‚ ‚éپB